Age Estimation
Last modified on Jan 21, 2021
Scale patterns can be interpreted to estimate fish age because scales grow incrementally and have patterns of rings, called circuli, which reflect fish growth rates (Fisher and Pearcy 1990). During periods of fast summer growth, circuli spacing is wider, and during slow winter growth, circuli spacing is narrower. Annuli, distinguished by wide (summer) then narrow (winter) circuli, are counted to estimate fish age.
Chinook salmon are the largest Pacific salmon and are some of the largest and fastest-growing fish in the world (Groot 2010). The large body size of Chinook salmon is due to rapid growth and prolonged life (Groot 2010). Because circuli spacing is not always different between summer and winter, identification of Chinook salmon annuli are particularly challenging (see examples in scale images by area).
Salmon ages are recorded in European notation (Koo 1955; Mosher 1969) that includes the number of years spent in fresh water after emergence from the gravel, a period, followed by the number of years spent in the ocean. The total age of the fish is the sum of the number of years spent in fresh water and the number of years in the ocean plus one to account for time spent in the gravel as an embryo.
On this page are descriptions of scale characteristics used to estimate age:
- Annuli
- Checks
- Freshwater growth
- Estuarine and ocean growth
- The final ocean annulus and "plus" growth
Auxiliary information is used to increase confidence and narrow the range of possible age estimates.
Annuli
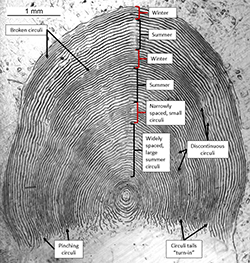
An annulus (plural, annuli) consists of one year of growth. The end of each year on a scale corresponds to the end of winter, and winters are counted to estimate age. Characteristics of winter growth include:
- Narrowly-spaced circuli relative to wider circuli spacing during summer growth (Figure 1).
- Small circuli relative to larger circuli during summer growth (Figure 1).
- Discontinuous, broken circuli (i.e. circuli that are incomplete from one side of the scale to the other, Figure 1).
- Braiding and branching of circuli.
- Circuli tails "turn in" at base of scale (Figure 1).
- Extension of annulus into posterior portion of scale. This is visible via shadow, increased reticulation, or extension of circuli (Figure 2).
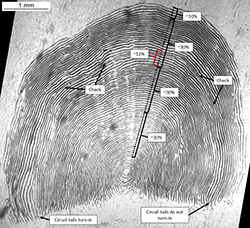
Annulus spacing, or distance between annuli, can be used to distinguish annuli. Visual cues for annulus spacing are:
- Seasonality of growth. Summer and winter seasons are apparent when circuli spacing is followed starting at the focus to the margin of a scale. There can be gradual widening of circuli spacing after winter growth and gradual compression of circuli spacing up to winter growth (Figure 3).
- Comparisons of the size of annuli. Annual growth zones tend to be similar in size. A check should be suspected if the growth zone is much smaller or much larger than adjacent annuli (Figure 3). Comparisons of counts of circuli within each annulus is another method for comparing relative size of annuli.
- Completeness of each annulus. Annuli can be followed from one end of circuli tails to the other.
Unfortunately, not all of these characteristics are present on every annulus and there is no single characteristic that can be used to identify all annuli. Also, there are periods of reduced growth that are not part annual growth, called checks, that can be confused with annuli because they have some of the same visual cues as annuli (see next section).
Note on growth pattern trends: Growing conditions vary from year to year, which can result in anomalous patterns of growth (proportionally wider or narrower annuli or persistent checks) consistent for a brood year. These can sometimes be recognized as belonging to a specific age and recognition of these patterns can help with estimating age (Tobias et al. 1994).
Checks
Periods of reduced growth due to something other than winter can leave "checks" on the scale. Checks can be mistaken for annuli because they have some of the a annulus features listed above. The following features can be used to distinguish checks from annuli:
- Checks are out of place within seasonal growth pattern.
- Relative to seasonal circuli growth: Circuli following a check may have abrupt, widely-spaced circuli before and after the check with no gradual increase in spacing. Seasonal growth patterns can be collected from other scales from the same fish or other fish from same population. As scales are read, familiarity with an expected seasonal pattern helps separate checks from annuli.
- Relative to the seasonal annuli growth: Spacing among annuli tend to be similar by age and population. Checks usually have a smaller distance to the closest annulus than between adjacent annuli.
- A check may be present on only a portion of the scale, whereas an annulus can be followed completely from one end of the circulus tails to the other.
- Circulus spacing within a check, although narrower than summer circuli spacing, may be wider than winter circuli spacing.
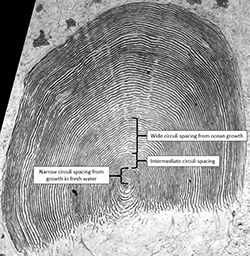
Freshwater growth
Salmon growth in fresh water is slow relative to growth in the ocean (Groot 2010), and this is apparent in scale circuli spacing. Freshwater circuli are narrowly-spaced and smaller than ocean circuli (Figure 4). The direction of circuli tails (ends of circuli) turning towards the focus has been used to identify freshwater growth and the tails turning out has been used to identify the beginning of growth in the ocean (ocean entrance; Figure 4; Bagliniere and Ombredane 1990).
The average size and circuli count of freshwater growth to the first winter annulus for Alaskan Chinook salmon is 0.35 mm and 11 circuli (SD = 0.02 and 0.85) for age 1.2 fish; 0.33 mm and 11 circuli (SD = 0.02 and 0.77) for age 1.3 fish; 0.33 mm and 11 circuli (SD = 0.02 and 0.88) for age 1.4 fish; and 0.34 mm and 10 circuli (SD = 0.03 and 1.16) for age 1.5 fish from the average measurements for 7, 17, 17, and 4 populations ranging from southeast to northwest Alaska (L. Wilson, personal communication, January 25, 2019). For comparison, Juvenile Chinook salmon in the upper Yukon River was less and ranged from 5-7 circuli (Murray 1990).
Freshwater scale growth that is after the winter but before estuarine or ocean entrance is freshwater "plus" growth. If there is a large amount of freshwater plus growth, the combination of freshwater plus growth and a transition between freshwater and estuarine or ocean growth can look like second freshwater annulus. Fish with longer migrations from freshwater to the ocean and substantial amounts of freshwater growth can lead to disagreements in assigning a second freshwater annulus (DuBois and Liller 2010).
Most Alaskan Chinook salmon have a stream-type life history. These fish rear in their home river from the summer after emergence to the following spring and will have one freshwater annulus (Groot 2010). Chinook salmon with ocean-type life histories emigrate to the ocean the summer after emergence and do not have a freshwater annulus. Scale pattern analysis revealed that the freshwater zone on scales from Chinook salmon with a stream-type life history have a compact band of closely spaced circuli that were similar between populations, whereas the freshwater zone size on scales from Chinook salmon with an ocean-type life history was smaller than for stream-type and variable among populations (Unwin and Lucas 1993). Scale circulus spacing and freshwater zone size for stream-type and each population of ocean-type population were consistent with fresh water growth and out-migrant size (Unwin and Lucas 1993).
The freshwater growth patterns on scales of ocean-type Chinook salmon may be difficult to distinguish from stream-type. For example, In southeast Alaska, Situk River Chinook salmon were originally classified as stream-type because freshwater annuli appeared to be present from looking at their scale patterns, although they do not. After Situk River Chinook salmon smolt timing was studied, they found that their smolt emigrate to the ocean after emergence and during the summer, thus they have an ocean-type life history (Johnson et al. 1992). This exemplifies the importance of studying the life history of the population of interest. Exposure to scales of differing life histories increases age estimation accuracy. For example, a reader with years of scale reading experience but limited exposure to other life histories had 84% accuracy, but a reader with years of reading experience and broad exposure to life history types had 95% accuracy (McNicol and MacLellan 2010).
Estuarine and ocean growth
Ocean and estuarine circuli are large and more widely spaced than freshwater circuli (Figure 5). Circuli formed while the fish was in the ocean cannot always be distinguished from those formed while in an estuary, with successful separation levels that vary among watersheds and populations. In Alaska, where most fish are stream-type and have only one freshwater annulus, intermediate growth that occurs after a fish has emigrated from freshwater but has narrower circuli spacing than those formed in mid-summer, can appear like a second freshwater annulus. In the Columbia River watershed, where tidal freshwater extends more than 230 km upriver from the mouth, checks and indeterminate growth on scales underestimated the estuarine residency of fish and fish with distinct scale checks sometimes had no chemical evidence of estuary entry on their scales or otoliths (Campbell et al. 2015). On the other hand, in the Sixes River, Oregon, where the estuary is small and tidal influence is limited, scale characteristics and growth transitions during downstream migration were closely-linked and scale checks accurately depicted estuary entrance (Reimers 1973).
Because of their robust growth, Chinook salmon winter growth consists of a few to >10 circuli. Circuli spacing can be the same between summer and winter growth, making identification of other winter growth characteristics valuable.
The final ocean annulus and "plus" growth
Alaska Chinook salmon are among the first salmon to return during spawning migration (Groot and Margolis 1991). Because they return to their natal river in the spring or summer, they have little to no growth after the final winter. The growth between the final winter and the scale margin is "saltwater plus growth."
The amount of saltwater plus growth increases with sample day. For example, a fish sampled early in the year (e.g.; 15 June) will show less plus growth than a fish sampled late in the year (1 August). The amount of plus growth decreases with number of years a fish spent in the ocean. For example, a fish that spent one year in the ocean generally will have more plus growth than a fish that spent four years in saltwater.
Few studies exist on the timing of ocean annuli for Chinook salmon. The month of final annulus formation differed by behavioral type for age 0.2, 0.3, and 1.3 Chinook salmon caught off Washington. Ocean-type Chinook salmon formed the final annulus in March, whereas stream-type Chinook formed the final annulus by April (Hyun et al. 1998).
References
Baglinière, E., and D. Ombredane. 1990. Choix et fiabilité d'un modèle de rétrocalcul des tailles en écologie halieutique. Mise au point d'un logiciel informatique. Bulletin Français de la Pêche et de la Pisciculture 319: 258-272.
Campbell, L. A., D. L. Bottom, E. C. Volk, and I. A. Fleming. 2015. Correspondence between scale morphometrics and scale and otolith chemistry for interpreting juvenile salmon life histories. Transactions of the American Fisheries Society 144(1): 55-67.
DuBois, L., and Z. W. Liller. 2010. Yukon River Chinook salmon aging consistency. Alaska Department of Fish and Game, Fishery Data Series No. 10-45, Anchorage. http://www.adfg.alaska.gov/FedAidpdfs/FDS10-45.pdf.
Fisher, J. P., and W. G. Pearcy. 1990. Spacing of scale circuli versus growth rate in young coho salmon. Fishery Bulletin 88: 637-643.
Groot, C. 2010. Physiological ecology of Pacific salmon. UBC Press.
Groot, C., and L. Margolis. 1991. Pacific salmon life histories. UBC press.
Hyun, S.-Y., K. W. Myers, and J. Sneva. 1998. The Time of Annulus Formation in Chinook Salmon Caught in Washington Coastal Waters. (NPAFC Doc. 352). Fisheries Research Institute, University of Washington, Seattle, WA.
Johnson, S., J. Thedinga, and K. Koski. 1992. Life history of juvenile ocean-type chinook salmon (Oncorhynchus tshawytscha) in the Situk River, Alaska. Canadian Journal of Fisheries and Aquatic Sciences 49(12): 2621-2629.
Koo, T. S.-y. 1955. Biology of the Red Salmon, Oncorhynchus Nerka (Walbaum) of Bristol Bay, Alaska as Revealed by a Study of Their Scales. University of Washington.
McNicol, R. E., and S. E. MacLellan. 2010. Accuracy of Using Scales to Age Mixed-Stock Chinook Salmon of Hatchery Origin. Transactions of the American Fisheries Society 139(3): 727-734.
Mosher, K. H. 1969. Identification of Pacific salmon and steelhead trout by scale characteristics. United States Department of the Interior, Fish and Wildlife Service, Bureau of Commercial Fishes.
Murray, C. B., M. A. Henderson, and T. D. Beacham. 1990. Size and Scale Characteristics of Upper Yukon River Juvenile Chinook Salmon (Oncorhynchus tschawytscha). Canadian Technical Report of Fisheries and Aquatic Sciences 1767. Nanaimo, British Columbia.
Reimers, P. E. 1973. The length of residence of juvenile fall Chinook salmon in Sixes River, Oregon. Fish Commission of Oregon.
Tobias, T. M., D. L. Waltemyer, and K. E. Tarbox. 1994. Scale aging manual for Upper Cook Inlet sockeye salmon. Alaska Department of Fish and Game, Commercial Fisheries Management and Development Division, Regional Information Report 2A94-36, Anchorage. http://www.adfg.alaska.gov/FedAidpdfs/RIR.2a.1994.36.pdf
Unwin, M. J., and T. P. Quinn. 1993. Homing and straying patterns of chinook salmon (Oncorhynchus tshawytscha) from a New Zealand hatchery: spatial distribution of strays and effects of release date. Canadian Journal of Fisheries and Aquatic Sciences 50(6):1168-1175.